Share:
Asante receives its first shipment of coronavirus vaccine
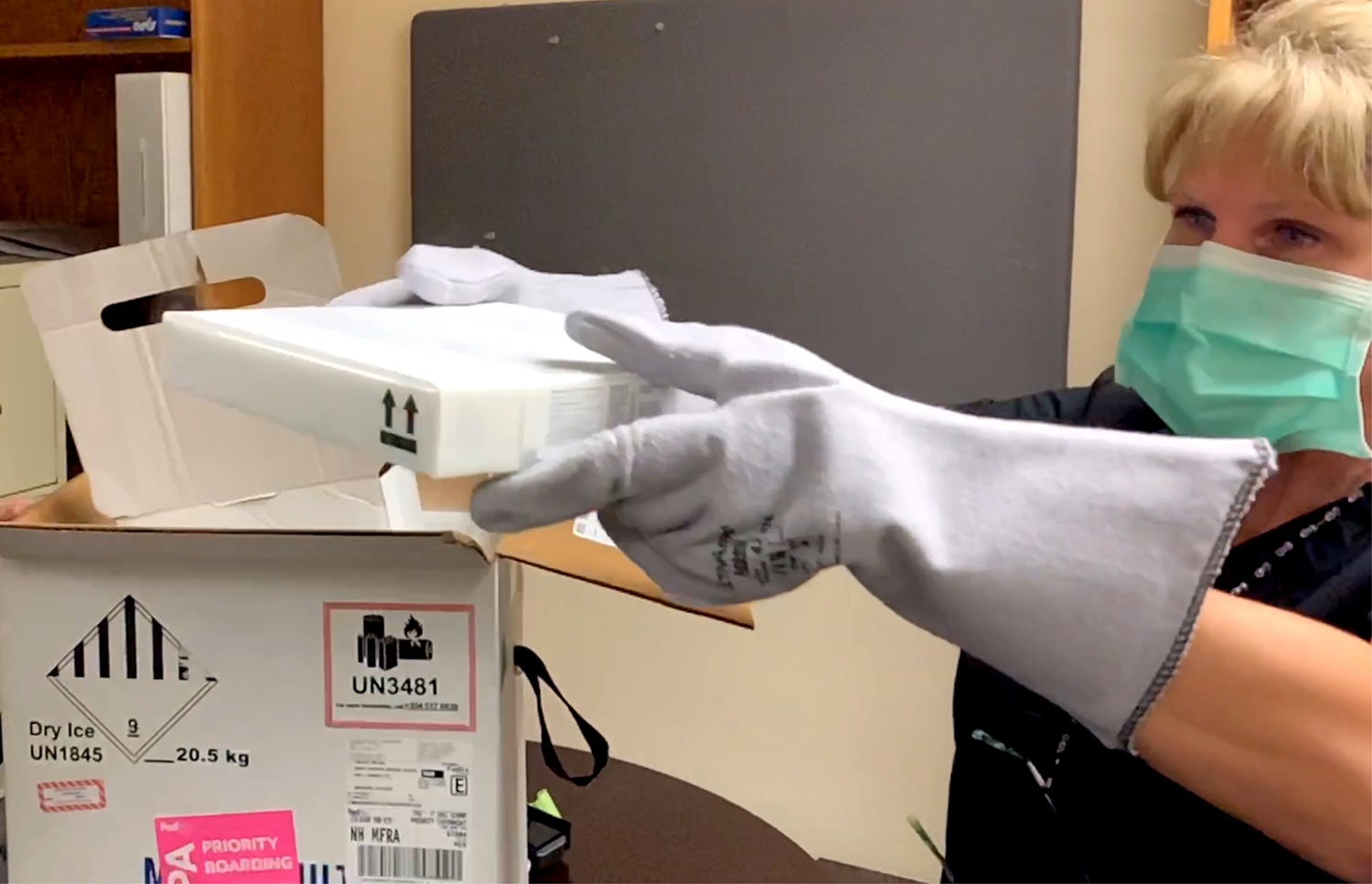
After eight long months, Asante employees are seeing light at the end of the tunnel of the COVID-19 pandemic. The first shipment of the Pfizer coronavirus vaccine arrived at Asante today and vaccinations begin tomorrow for select frontline workers.
The system expects to immunize all Asante employees who want the vaccine by the end of February, Holly Nickerson, vice president of Quality, said at a news conference this morning.
Asante is among the first five health systems in the state to receive a vaccine. The initial shipment covers less than 15% of Asante’s workforce, so clinical leaders are prioritizing which job functions will receive the vaccine.
Eligibility is ranked by tiers, with departments at the highest risk of exposure at the top. Tier One departments include:
- Emergency
- Respiratory urgent care
- Areas involving aerosol-generating procedures
- Med Surg hot zones
- Critical care units
Job categories within those Tier One departments are credentialed providers; nurses, CNAs and techs; physical, occupational, speech/language, and respiratory therapists; phlebotomists, EVS staff and imaging staff.
Employees in Tier One departments and job categories should call Asante Employee Health to schedule an appointment. All other staff will receive notification when they are eligible to schedule an appointment as more shipments arrive.
Vaccinations are given in two doses, three weeks apart and tracked in the employee’s health record. Shots will be administered in a Medford location, which is not being released publicly. Employees will receive the name and address of the location when they make their appointment.
For now, Asante is using only the Pfizer vaccine, which requires the ultracold storage available locally only at Asante. This allows other vaccines, such as one being produced by Moderna, to be distributed at long-term care facilities and other rural providers.
Physicians who volunteered for the vaccine’s national clinical trials reported some mild symptoms after receiving their first and second doses. These include fatigue, headache and swollen lymph nodes, indicating an effective immune response, said Courtney Wilson, MD, vice president of Medical Affairs at ARRMC.
In a recent systemwide survey, about 60% of Asante employees said they wanted the vaccine; others wanted to wait or said they would decline.
As with flu immunization, the coronavirus vaccine is strongly encouraged, but not mandatory at Asante. Clinical studies have found the Pfizer vaccine to be 95% effective and the Moderna vaccine 94.5% effective. The Pfizer vaccine was granted emergency use authorization by the FDA, with the Moderna vaccine expected to follow this week.
“As the region’s largest health care provider, we owe it to our patients and our community to deliver safe and effective care,” said Jamie Grebosky, MD, Asante’s chief medical and quality officer. “Immunizing our staff is an important first step to help stem this pandemic.”
If you have a question, please contact the author or relevant department directly.




3 Comments. Leave new
The online appts. for second Pfizer shot are all full for 2/15, 2/16 & 2/17. How may I schedule an appt. during my recommended window?
@MJ Friedrich-Sanders – Please request your booster at an Asante Vaccine Clinic on this page: https://www.asante.org/covid-19/vaccine-clinic/request-vaccination/
Our vaccination scheduling team will contact you within one week, give you a choice of vaccination clinic locations and discuss appointment availability.
How do I register for the second Merdana vaccine shot. Thanks